Waves
wave: energy travelling from one place to another.
medium: the substance that a wave travels through. Electromagnetic waves (including light) can travel without a medium.
· The wave of energy travels through the medium.
· All (or nearly all) of the energy passes through the medium—the medium doesn’t absorb it.
Some examples of waves:
|
Type of Wave |
Medium |
|
sound |
air (or water, solids) |
|
ocean |
water |
|
electromagnetic (e.g., light, radio) |
anything (even vacuum) |
Types of waves:
transverse wave: moves its medium up & down (or back & forth) as it travels through. Examples: light, ocean waves

longitudinal
wave (or compressional wave): compresses and decompresses the medium as it
travels through.
Examples: sound, pressure.
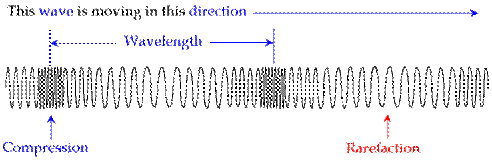
wavelength (or period): the length of the wave, measured from a specific point in the wave to the same point in the next wave. Symbol = λ (lambda); unit = distance
frequency: the number of waves that travel past a point in a given time. Symbol = ν (nu); unit = 1/time
speed = λν
spectrum: the set of all possible wavelengths. Visible light is one set of wavelengths that are part of the full electromagnetic spectrum.
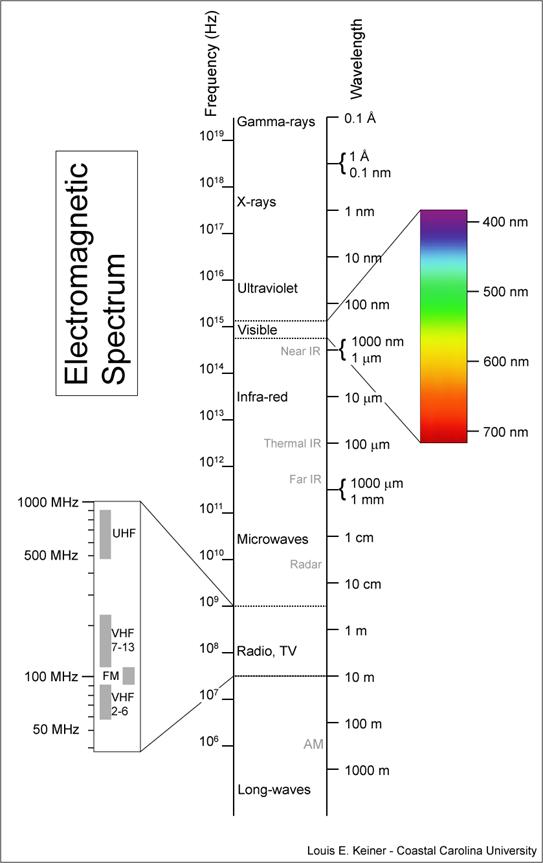
Electromagnetic waves (such as light, radio waves, etc.) travel at a constant speed—the speed of light. The speed of light is a constant, and is denoted by the letter “c” in equations.
c = 3.00 × 108 m/s = 186,000 miles per second
The energy (E) that a wave carries equals a constant times the frequency. (Think of it as the number of bursts of energy that travel through the wave every second.) For electromagnetic waves (including light), the constant is Planck’s constant (named after the physicist Max Planck), which is denoted by a script h in equations. So the equation is:
E = hν
h = 6.63 ×10−34 J∙s
Louis de Broglie: French physicist. Showed that any object with momentum (i.e., has mass and is moving) creates a wave as it moves. Large objects with a lot of momentum (such as people) create waves with wavelengths that are too small to detect. Small objects (such as electrons) create waves with wavelengths in the visible part of the spectrum, which are easy to detect.